In the beginning of this project, we did many labs and experiments to familiarize ourselves with the 5 common types of chemical reactions (Single replacement, Double replacement, Decomposition, Synthesis, and Combustion) to familiarize ourselves with them. We were then expected to, in groups of four, design and build a themed board game that featured some of these reactions. We ended up making a board game with an under the sea theme, called Chemisea, that had five different reactions. The first reaction was the pufferfish, which used hydrochloric acid combined with calcium carbonate. This double replacement reaction resulted in carbon dioxide, which we used to inflate a balloon with a pufferfish drawn on it. Our second reaction was a squid ink reaction, which was another double replacement reaction. it used silver nitrate and potassium chromate, which, when combined, form silver chromate and potassium nitrate. Silver chromate has a nice deep red color, that looked like squid ink. The next reaction was our anglerfish. The anglerfish had an LED, which we lit using a penny battery. To make a penny battery, first sand down one side of a penny, until the zinc under the copper is visible. Then stack alternating pennies and acid soaked cardboard disks, until the stack can generate enough electricity to light an LED. I was in charge of the penny battery, and had a decent amount of success. Our next reaction was the whale, which used a reaction commonly known as "elephant toothpaste". Elephant toothpaste is a decomposition reaction, where hydrogen peroxide combined with soap slowly forms water and oxygen. The catalyst potassium iodide is used to speed up the process, and cause the whale to blow foam out of its blowhole. Our final reaction was the underwater volcano. We used a Bunsen burner in our volcano, which is a combustion reaction. The methane gas the burner uses combines with oxygen in the air, resulting in carbon dioxide and water vapor. We put cupric chloride into the fire, which made it burn green. Our game itself played similar to Candyland, where the players drew cards to move across the board. There were shortcuts, and tiles with red x's on them. When a player ended their turn on a red x tile, the reaction associated with the tile was triggered (if it had not already been), and the player was moved back one tile. The game ended when a player reached the treasure chest at the end of the path.
Concepts Used:
Atom: An atom is the building block for everything. Everything on Earth is made of atoms. Atoms are formed from protons, neutrons, and electrons.
Proton: Protons are one of the three molecules that form atoms. They can be found in the nucleus of the atom, and help determine the atom's mass. They have a charge of +1, and the number of protons an atom has determines it's atomic number.
Neutrons: Neutrons are one of the three molecules that form atoms. They can be found in the nucleus of the atom, and help determine the atom's mass. They have a charge of 0, and the number of neutrons and protons an atom has determines it's mass.
Electrons: Electrons are one of the three molecules that make up an atom. Electrons orbit around the nucleus, forming most of the volume of the atom, and they have almost no mass. Electrons have a charge of -1. The number of electrons compared to the number of protons determines of an atom has a positive charge, a negative charge, or no charge.
Element: An element is a piece of matter that is made up of only one type of atom. An example would be iron, which is made up of only iron atoms.
Compound: A compound is a molecule made up of multiple elements, so it has more than one type of atom. An example would be acetate, which is made of two carbon, three hydrogen, and two oxygen atoms.
Cation: A cation is an element or compound that has more protons than electrons. Metals are almost always cations.
Anion: An element or compound that has more electrons than it has protons. Anions have a negative charge. Non-metals are generally anions, with the notable exceptions of ammonium and hydronium.
Single Replacement Reaction: A single replacement reaction is when a element swaps places with either the anion or cation portion of a compound. This takes the form of A+BC = B+AC, or A+BC = C+BA, depending on whether the element is a cation or an anion.
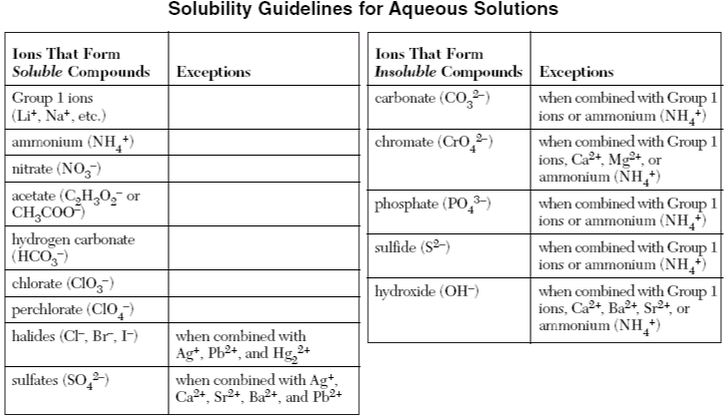
Double Replacement Reaction: A double replacement reaction is when two compounds in aqueous solution swap either cations or anions. This reaction only occurs if one of the compounds formed will be insoluble, according to the guidelines below.
Atom: An atom is the building block for everything. Everything on Earth is made of atoms. Atoms are formed from protons, neutrons, and electrons.
Proton: Protons are one of the three molecules that form atoms. They can be found in the nucleus of the atom, and help determine the atom's mass. They have a charge of +1, and the number of protons an atom has determines it's atomic number.
Neutrons: Neutrons are one of the three molecules that form atoms. They can be found in the nucleus of the atom, and help determine the atom's mass. They have a charge of 0, and the number of neutrons and protons an atom has determines it's mass.
Electrons: Electrons are one of the three molecules that make up an atom. Electrons orbit around the nucleus, forming most of the volume of the atom, and they have almost no mass. Electrons have a charge of -1. The number of electrons compared to the number of protons determines of an atom has a positive charge, a negative charge, or no charge.
Element: An element is a piece of matter that is made up of only one type of atom. An example would be iron, which is made up of only iron atoms.
Compound: A compound is a molecule made up of multiple elements, so it has more than one type of atom. An example would be acetate, which is made of two carbon, three hydrogen, and two oxygen atoms.
Cation: A cation is an element or compound that has more protons than electrons. Metals are almost always cations.
Anion: An element or compound that has more electrons than it has protons. Anions have a negative charge. Non-metals are generally anions, with the notable exceptions of ammonium and hydronium.
Single Replacement Reaction: A single replacement reaction is when a element swaps places with either the anion or cation portion of a compound. This takes the form of A+BC = B+AC, or A+BC = C+BA, depending on whether the element is a cation or an anion.
Double Replacement Reaction: A double replacement reaction is when two compounds in aqueous solution swap either cations or anions. This reaction only occurs if one of the compounds formed will be insoluble, according to the guidelines below.
Synthesis Reaction: A synthesis reaction is when two or more elements or compounds all combine to form only one product. If a reaction has a single product, it must be a synthesis reaction.
Decomposition Reaction: A decomposition reaction is when one compound breaks apart into two or more elements or compounds. It is basically the opposite of a synthesis reaction, with only one reactant and multiple products.
Decomposition Reaction: A decomposition reaction is when one compound breaks apart into two or more elements or compounds. It is basically the opposite of a synthesis reaction, with only one reactant and multiple products.
Reflections:
During this project, I did some things well. One thing I did well was outside work. Throughout this project, I was working on things at home, be it building the anglerfish or making the penny battery. I was willing to put in the extra effort, and I am proud of that. Another thing I succeeded at in this project was flexibility. I worked with my team, and capitulated to their requests, instead of arguing my points.
There were some points at which I failed during this project, however. One of those points was communication. I had a hard time communicating what I thought was necessary to my team at times, though not always. Another failing point was time management. I felt extremely rushed near the end of the project, and I think that if I had managed my time better, this wouldn't have happened.
During this project, I did some things well. One thing I did well was outside work. Throughout this project, I was working on things at home, be it building the anglerfish or making the penny battery. I was willing to put in the extra effort, and I am proud of that. Another thing I succeeded at in this project was flexibility. I worked with my team, and capitulated to their requests, instead of arguing my points.
There were some points at which I failed during this project, however. One of those points was communication. I had a hard time communicating what I thought was necessary to my team at times, though not always. Another failing point was time management. I felt extremely rushed near the end of the project, and I think that if I had managed my time better, this wouldn't have happened.