In this unit, we as 8 separate groups of 4, each made a design for the most energy-efficient outdoor-area. Our plans were then reviewed by a board of architects and engineers, who selected the best plan. This plan was then built on campus. This was a very long project, so I will sum up each of the major things we did with it's own paragraph.
Part I: Atomic Structure
For this part, we learned about atoms, and their structures. Atoms have three basic parts: protons, neutrons, and electrons. The protons and neutrons are in the center of the atom, and they have make up the vast majority of the atom's mass. The interesting thing is is that the nucleus of the atom (both protons and neutrons) is only 1/100,000 of the atoms size. This is because the atom's vast majority is empty space, with only a few electrons orbiting the nucleus. The electrons do not actually orbit the nucleus, however. They simply seem to teleport around the nucleus, appearing at random places in the designated areas that they can be in. (This description may not be entirely accurate, but since on this scale, things happen that we just cannot explain, going further could melt your brain). Besides the composition of atoms, we also learned about their state and heat. Since these two concepts are mixed together, I will discuss them both. A materials state is determined by it's heat. After an object absorbs enough energy, (perceived by us as heat), it's atoms begin moving fast enough that they can no longer form the same kinds of bonds. When materials have the least energy, they are solids, characterized by having strong bonds that don't change. Atoms move, but not enough to break the bonds. After the material absorbs a sufficient amount of energy (this amount varies), it becomes a liquid. Liquids atoms bond with each other, but they have enough energy to break these bonds and form new ones. The final state, achieved after absorbing even more energy, is gas. The atoms of a gas have so much energy that they very rarely form bonds, and when they do, the bonds don't last long. A gases atom's are simply moving too fast.
For this part, we learned about atoms, and their structures. Atoms have three basic parts: protons, neutrons, and electrons. The protons and neutrons are in the center of the atom, and they have make up the vast majority of the atom's mass. The interesting thing is is that the nucleus of the atom (both protons and neutrons) is only 1/100,000 of the atoms size. This is because the atom's vast majority is empty space, with only a few electrons orbiting the nucleus. The electrons do not actually orbit the nucleus, however. They simply seem to teleport around the nucleus, appearing at random places in the designated areas that they can be in. (This description may not be entirely accurate, but since on this scale, things happen that we just cannot explain, going further could melt your brain). Besides the composition of atoms, we also learned about their state and heat. Since these two concepts are mixed together, I will discuss them both. A materials state is determined by it's heat. After an object absorbs enough energy, (perceived by us as heat), it's atoms begin moving fast enough that they can no longer form the same kinds of bonds. When materials have the least energy, they are solids, characterized by having strong bonds that don't change. Atoms move, but not enough to break the bonds. After the material absorbs a sufficient amount of energy (this amount varies), it becomes a liquid. Liquids atoms bond with each other, but they have enough energy to break these bonds and form new ones. The final state, achieved after absorbing even more energy, is gas. The atoms of a gas have so much energy that they very rarely form bonds, and when they do, the bonds don't last long. A gases atom's are simply moving too fast.
|
Part II: Solar Hot Water
For this part, we designed a machine to heat water using only solar power. We learned about heat transfers, insulators, and conductors to do so. there are three kinds of heat transfers: conduction, convection, and radiation. Conduction is the transfer of heat through solids. An example of this is when a cold floor sucks the heat out of your feet. Convection is the transfer of heat through fluids. An example is stepping into a warm room. Radiation is the transfer of heat through waves, rays, or particles. An example of this is the sun's rays warming the earth. Insulators are materials that absorb heat very slowly. Since trapped air is a very good insulator, most insulators use trapped air. An example is styrofoam. Conductors are the opposite of insulators. They are materials that absorb and transfer heat very quickly. Most metals are good conductors, with copper being one of the best conductors. Using all of this knowledge, we designed a solar hot water heater. It used tinfoil to reflect the sun's rays onto a copper pipe containing water. It was not very effective, because we failed to caulk one of the connections properly. |
|
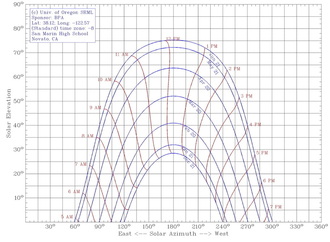
Part III: Solar Angles
For this part, we learned about the how the angle of the Earth affects the seasons. When the sun's rays hit a tilted part of the Earth, the rays are spread out over a greater area. Since the sun always puts out the same amount of rays, when the area is increased, the density decreases. This means that when the rays are spread over a greater area, each part gets less heat, making it winter. |
Part IV: Client Needs Survey
For this part, we decided who would use an outdoor class-room, and surveyed them on what they wanted in an outdoor class-room. We designed the survey as a class. While the survey's results were slightly skewed, due to our interviewing more students than teachers, the results were still quite interesting. We learned that people wanted an amphitheater and group-work layout for the class-room. We also discovered that The most widely request item by far was WiFi, but that this was mostly by students. The teachers most asked for item was a whiteboard.
For this part, we decided who would use an outdoor class-room, and surveyed them on what they wanted in an outdoor class-room. We designed the survey as a class. While the survey's results were slightly skewed, due to our interviewing more students than teachers, the results were still quite interesting. We learned that people wanted an amphitheater and group-work layout for the class-room. We also discovered that The most widely request item by far was WiFi, but that this was mostly by students. The teachers most asked for item was a whiteboard.
|
Part V: Site selection
In this part, we decided where our proposed class-room was going to be. To do this, we created a list of what we thought was important to have in a site. We then rated multiple sites based on these attributes. When the rankings for all the attributes were finished, we found that a site behind the library building had won. Unfortunately, we then found out that we couldn't actually build there. We ended up selecting the second best site, which was behind the art building near the tennis courts. You can see a picture of our sight on the right. |
Part VI: Materials Testing
In this part, we tested multiple materials for their ability to hold and retain heat, and their ability to absorb water. This was so we could determine what we wanted to make the structures in our outdoor class-room out of. We tested the object two ways. In the first part, we put the materials under heat lamps and took their temperature every five minutes. For the second part, we soaked objects in water, let them dry for five minutes, then took their weight to see how much water was left in them. We only tested wood for the second part. You can click the button below to see the data we collected.
In this part, we tested multiple materials for their ability to hold and retain heat, and their ability to absorb water. This was so we could determine what we wanted to make the structures in our outdoor class-room out of. We tested the object two ways. In the first part, we put the materials under heat lamps and took their temperature every five minutes. For the second part, we soaked objects in water, let them dry for five minutes, then took their weight to see how much water was left in them. We only tested wood for the second part. You can click the button below to see the data we collected.
|
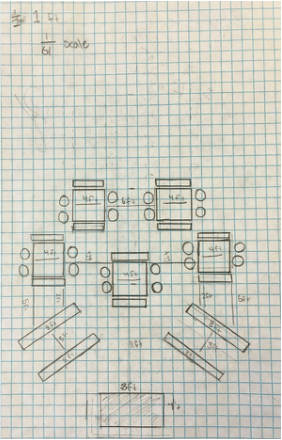
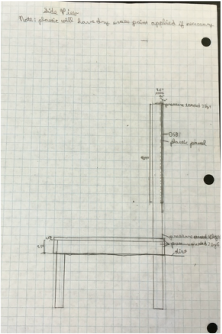
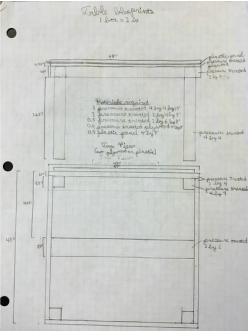
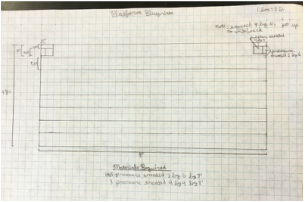
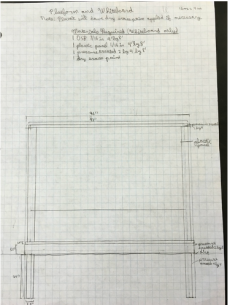
Part VII: Building Design
For this part, we designed the layout and structures we wanted to include in our outdoor classroom. We decided that the best layout was a stage, including a whiteboard, with two sets of two long benches in front of it, with five large tables behind the benches. You can see the general layout at right. Due to a spending limit of $3000, we decided to make everything out of pressure treated wood, which doesn't rot and is fairly cheap. The only it fails at is appearance, but that isn't really an issue. We then had to make blueprints of all our structures, which can be seen below. Our final step was building a scale model of one of our tables, so people could see how we had thought through how it would fit together. |
Part VIII: Justification
In this part, we as a class learned about all the different ways to power things to help us understand why we might want to make a classroom off the grid. My group covered nuclear power, which was fascinating. I found it interesting that nuclear power has claimed so many less lives than coal, it is still considered more dangerous. The document we assembled as a class that lists the pros and cons of each power source, as well as some other facts, can be found below.
In this part, we as a class learned about all the different ways to power things to help us understand why we might want to make a classroom off the grid. My group covered nuclear power, which was fascinating. I found it interesting that nuclear power has claimed so many less lives than coal, it is still considered more dangerous. The document we assembled as a class that lists the pros and cons of each power source, as well as some other facts, can be found below.
Part IX: Wind Turbine Lab
For this final part, we designed wind turbine blades in groups of four. We then constructed our designs out of paper or sheet metal, and tested them using fans and a leaf blower. After we had tested many designs, we were asked to make a hypothesis, and defend it to the class. Our hypothesis was that a large bladed vertical axis wind turbine with an odd number of blades would be the best. We believed this because large blades would catch the wind the best, and an odd number of blades would prevent the design from stalling out due to it being blown in both directions at once.
For this final part, we designed wind turbine blades in groups of four. We then constructed our designs out of paper or sheet metal, and tested them using fans and a leaf blower. After we had tested many designs, we were asked to make a hypothesis, and defend it to the class. Our hypothesis was that a large bladed vertical axis wind turbine with an odd number of blades would be the best. We believed this because large blades would catch the wind the best, and an odd number of blades would prevent the design from stalling out due to it being blown in both directions at once.
Concepts Used:
Heat: Heat is energy due to the movement of molecules or particles. It is represented by Q, and it's equation is mct. The "t" represents change in temperature, and the "c" represents specific heat (shown below). It is measured in Joules.
Specific heat: Specific heat is a constant for each material indicating how long it takes to heat up or cool down. It is measured in Joules per gram degree Celsius, and is represented by c. The higher c a material has, the more heat it can hold, and the slower it heats up.
Zeroth law of Thermodynamics: The first of the thermodynamic laws, this law states that if two systems are exchanging heat between each other, say systems A and B, and system B is also sharing heat with C, then system A is sharing heat with system C. A good way to think about this is what happens if you stick a metal pipe in the snow. You're hand will exchange heat with the snow, even though you aren't exactly touching it.
First law of Thermodynamics: The second of the thermodynamic laws, this law talks about how energy can never be destroyed it only changes form. This means that energy is only ever transferred from object to object, never created or destroyed.
Second law of Thermodynamics: The third of the thermodynamic laws, this laws states that over time, an isolated system will grow gradually more disordered. This law can be shown in a house, where when everyone is done with any object, they will simply drop it wherever they are. Over time, the house will become increasingly more disordered, until you reach the point where there is a very high chance that you will be able to find whatever you wanted wherever you are in the house, as long as the house began with enough of what you wanted.
Third law of Thermodynamics: The fourth of the thermodynamic laws, this law talks about how when a system approaches 0K, it's entropy gradually goes down.
Heat: Heat is energy due to the movement of molecules or particles. It is represented by Q, and it's equation is mct. The "t" represents change in temperature, and the "c" represents specific heat (shown below). It is measured in Joules.
Specific heat: Specific heat is a constant for each material indicating how long it takes to heat up or cool down. It is measured in Joules per gram degree Celsius, and is represented by c. The higher c a material has, the more heat it can hold, and the slower it heats up.
Zeroth law of Thermodynamics: The first of the thermodynamic laws, this law states that if two systems are exchanging heat between each other, say systems A and B, and system B is also sharing heat with C, then system A is sharing heat with system C. A good way to think about this is what happens if you stick a metal pipe in the snow. You're hand will exchange heat with the snow, even though you aren't exactly touching it.
First law of Thermodynamics: The second of the thermodynamic laws, this law talks about how energy can never be destroyed it only changes form. This means that energy is only ever transferred from object to object, never created or destroyed.
Second law of Thermodynamics: The third of the thermodynamic laws, this laws states that over time, an isolated system will grow gradually more disordered. This law can be shown in a house, where when everyone is done with any object, they will simply drop it wherever they are. Over time, the house will become increasingly more disordered, until you reach the point where there is a very high chance that you will be able to find whatever you wanted wherever you are in the house, as long as the house began with enough of what you wanted.
Third law of Thermodynamics: The fourth of the thermodynamic laws, this law talks about how when a system approaches 0K, it's entropy gradually goes down.
Reflections:
I fell that I did pretty well overall in this unit. One thing I did well was staying on track. I was able to keep working through all but the most severe distractions, and I am proud of that. I think I had a real improvement from my last project.
Sadly, one thing I didn't do well was keep my teammates on track. Some of them, especially Quinton, were of doing random things instead of our assigned work. I tried to get them to do things on my own, but I needed another teammate to get them to listen. I will have to work on being commanding.
Another thing I did well was my blueprints. I handled most of the blueprints, and I found that I really enjoyed it for some reason. It was calming to just focus on doing something I'm good on, drawing lines and fitting things together. I will explore this more in the future.
One final thing I did badly was agreeing with my teammates. I argued against a lot of their ideas, and the ideas were actually pretty good. I just didn't think they were better than my ideas, or I saw one small flaw, or something else. In the future I will have to work on accepting other's ideas.
I fell that I did pretty well overall in this unit. One thing I did well was staying on track. I was able to keep working through all but the most severe distractions, and I am proud of that. I think I had a real improvement from my last project.
Sadly, one thing I didn't do well was keep my teammates on track. Some of them, especially Quinton, were of doing random things instead of our assigned work. I tried to get them to do things on my own, but I needed another teammate to get them to listen. I will have to work on being commanding.
Another thing I did well was my blueprints. I handled most of the blueprints, and I found that I really enjoyed it for some reason. It was calming to just focus on doing something I'm good on, drawing lines and fitting things together. I will explore this more in the future.
One final thing I did badly was agreeing with my teammates. I argued against a lot of their ideas, and the ideas were actually pretty good. I just didn't think they were better than my ideas, or I saw one small flaw, or something else. In the future I will have to work on accepting other's ideas.
Below is my group's slideshow.