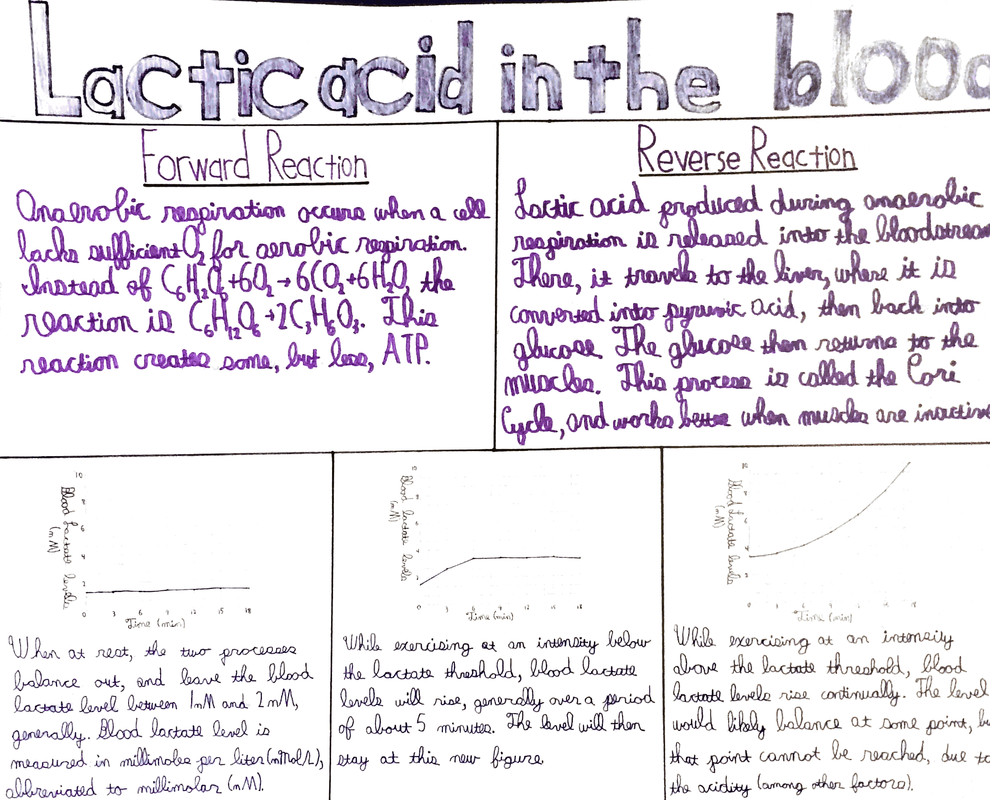
At the beginning of this unit, each students picked a topic that they cared about, or were interested in. We then individually researched our topics, finding credible sources that explained how the system was in equilibrium, and what could disrupt that equilibrium. After we had found all the information we needed, and created an annotated bibliography on our sources (which can be seen below), we created some form of product that would inform at least 100 others about what we had learned. The most common choice was a poster (which I chose as well), but there were also presentations, and posts on various social media sites. The subject I chose was lactic acid levels in the blood, which is controlled by anaerobic respiration in the muscles, and the Cori Cycle in the liver.
Concepts Used:
Reversible Reaction: A reaction is considered reversible when its products can react with each other to become its reactants again. Such reactions technically never end, only reach equilibrium. The symbol for a reversible reaction is ⇌.
Equilibrium: A reversible reaction has reached equilibrium when there is a stable, unchanging amount of both products and reactants. For example if there was a reaction, A ⇌ B, where the forward reaction rate (rate at which A becomes B) is 25%, the reverse reaction rate is 50%, an the reaction starts with 10 moles of A. After a while, the reaction will eventually have 6.66 moles of A, and 3.33 moles of B, and will not change from there. This occurs because every minute, 1.665 moles of A become B, and 1.665 moles of B become A. If the reaction rates or moles of products/reactants were to change, so would the equilibrium.
Le Chatelier's Principle: Le Chatelier's Principle dictates how a system in equilibrium will react to stress, namely, by moving away from it to reach equilibrium once more. A stress can be seen as the addition of product or reactant, changing of temperature, increasing of pressure, and other things beside. If the stress to the system occurs on the reactant's side, the equilibrium will shift to include more product, and the reverse holds true as well. To determine which side an increase in temperature stresses, look for the side which needs heat to work. To determine the same for a pressure increase, look to the side of the equation that has more molecules.
Reversible Reaction: A reaction is considered reversible when its products can react with each other to become its reactants again. Such reactions technically never end, only reach equilibrium. The symbol for a reversible reaction is ⇌.
Equilibrium: A reversible reaction has reached equilibrium when there is a stable, unchanging amount of both products and reactants. For example if there was a reaction, A ⇌ B, where the forward reaction rate (rate at which A becomes B) is 25%, the reverse reaction rate is 50%, an the reaction starts with 10 moles of A. After a while, the reaction will eventually have 6.66 moles of A, and 3.33 moles of B, and will not change from there. This occurs because every minute, 1.665 moles of A become B, and 1.665 moles of B become A. If the reaction rates or moles of products/reactants were to change, so would the equilibrium.
Le Chatelier's Principle: Le Chatelier's Principle dictates how a system in equilibrium will react to stress, namely, by moving away from it to reach equilibrium once more. A stress can be seen as the addition of product or reactant, changing of temperature, increasing of pressure, and other things beside. If the stress to the system occurs on the reactant's side, the equilibrium will shift to include more product, and the reverse holds true as well. To determine which side an increase in temperature stresses, look for the side which needs heat to work. To determine the same for a pressure increase, look to the side of the equation that has more molecules.
Reflections:
As this was not a group project, I was better able to see how I do on my own. I believe that I was good at finding sources with relevant information. Often it can be difficult to locate the facts required for such a project, but this time, I located my information in an acceptable amount of time. Another success in this project was my focus. I managed to stay on track and productive during all of the allotted work time, even without others pushing me to do so. I didn't succumb to the temptations of anything during my work.
There were still some failings during my work, however. first, I didn't manage my time very well. When I heard there were 3 work weeks for this project, I spent the first week doing minimal work. This culminated in me having to rush a little at the very end. Hopefully, I can avoid such procrastination in the future. Secondly, my poster-making skills were not very well honed. My poster wasn't exactly pleasing to look at, and while it wasn't the worst poster I saw, it certainly wasn't the best either.
As this was not a group project, I was better able to see how I do on my own. I believe that I was good at finding sources with relevant information. Often it can be difficult to locate the facts required for such a project, but this time, I located my information in an acceptable amount of time. Another success in this project was my focus. I managed to stay on track and productive during all of the allotted work time, even without others pushing me to do so. I didn't succumb to the temptations of anything during my work.
There were still some failings during my work, however. first, I didn't manage my time very well. When I heard there were 3 work weeks for this project, I spent the first week doing minimal work. This culminated in me having to rush a little at the very end. Hopefully, I can avoid such procrastination in the future. Secondly, my poster-making skills were not very well honed. My poster wasn't exactly pleasing to look at, and while it wasn't the worst poster I saw, it certainly wasn't the best either.